 So how good is our atomic radius? Standard tables give the atomic radius of strontium is in the range 192-220 pm. Get control of 2022 Track your food intake, exercise, sleep and meditation for free. Molar mass of C is 12.01070 ± 0.00080 g/mol. Molar mass of C is 12.01070 0.00080 g/mol Compound name is carbon Get control of 2022 Track your food intake, exercise, sleep and meditation for free. 99.0 atom 13C, <3 atom 18O CAS Number: Molecular Weight: 45.00 Beilstein: 3087932 MDL number: MFCD00064513 PubChem Substance ID: 24862443. Solution Molecules of this compound are composed of 13 carbon atoms, 18 hydrogen atoms, and 2 oxygen atoms. Since 3 3=27 and 4 3 = 64, you know that the cube root of 55 will be between 3 and 4, so the cube root should be a bit less than 4 × 10 –8. Molar Mass, Molecular Weight and Elemental Composition Calculator. 'Molecular formula' 'C'n'H'(n r) If you have heteroatoms, you adjust the formula. Element: Carbon Symbol: C Atomic Mass: 12.0107 of Atoms: 1 Mass Percent: 100.000 Calculate the molecular weight of a chemical compound Enter a chemical formula: Browse the list of common chemical compounds. Variations in the n(13C)/n(12C) ratio of terrestrial sources of carbon are caused largely. Average atomic mass of carbon is 12.011 what is the abundance of C12 in nature 14624066. > The formula is used in interpreting mass spectra. Percentages can be entered as decimals or percentages (i.e. Enter an optional molar mass to find the molecular formula. To calculate the empirical formula, enter the composition (e.g.  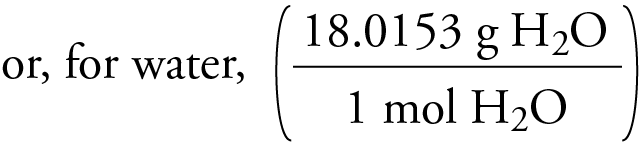  
Molar mass unit (g/mol) Molar mass is a macroscopic (large scale) unit of masses of atoms or molecules. The rule of 13 states that the formula of a compound is a multiple n of 13 (the molar mass of 'CH') plus a remainder r. This program determines both empirical and molecular formulas. \right ) \right ] \]ĭalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers.\), for example. The value of 1 a.m.u is 1.66 x 10 -24 g which is exactly the mass of 1 mole of carbon atoms. How to find the Molar Mass of CO: Carbon monoxide Wayne Breslyn 638K subscribers Subscribe 6.9K views 1 year ago Explanation of how to find the molar mass of CO: Carbon monoxide. Solution: A The molecular formula of ethanol may be written in three different ways: CH 3 CH 2 OH (which illustrates the presence of an ethyl group, CH 3 CH 2, and an OH group), C 2 H 5 OH, and C 2 H 6 O all show that ethanol has two carbon atoms, six hydrogen atoms, and one oxygen atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
